
Our Approach

Hormonal Changes and Symptom Burden in Menopause
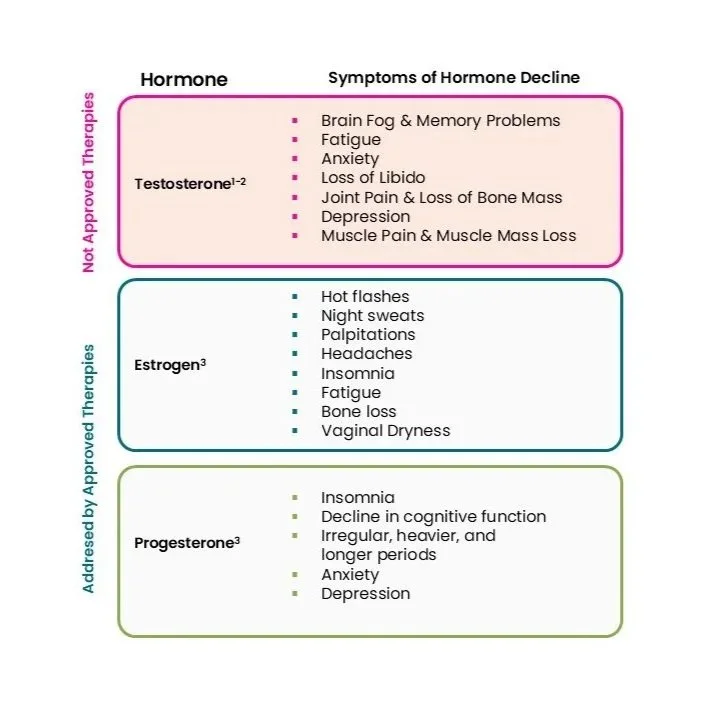
The hormonal changes associated with menopause are linked to a range of symptoms, including fatigue, mood changes, sleep disturbance, vasomotor symptoms, and musculoskeletal discomfort.
These symptoms vary across individuals in both severity and underlying drivers, reflecting the complex physiological changes that occur with loss of ovarian function.
Hypoandrogenism in Women
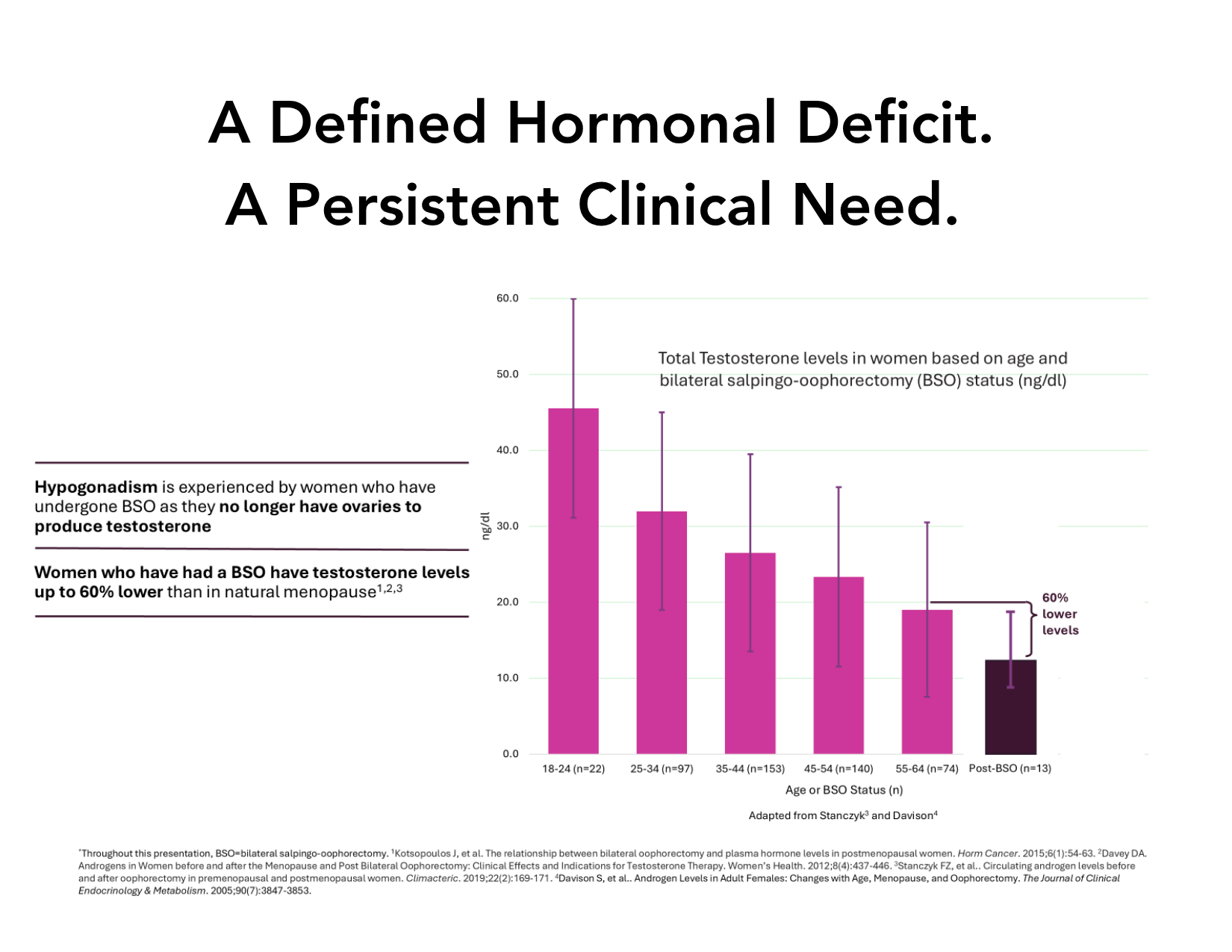
Hypoandrogenism refers to a state of reduced androgen activity and may occur in the setting of declining ovarian function or following oophorectomy, where loss of ovarian androgen production is more pronounced.
In some women, reduced androgen exposure is associated with clinically meaningful symptoms, particularly in domains such as sexual function and overall well-being.
Long-Term Health Considerations
The hormonal changes associated with menopause are also linked to longer-term health effects, including changes in bone density, body composition, and metabolic function.
While estrogen plays a central role in maintaining bone health, androgens contribute to musculoskeletal integrity and may influence bone density, muscle mass, and physical function.
Loss of ovarian function, particularly following oophorectomy, results in reduced exposure to both estrogen and androgen, which together may contribute to these long-term physiological changes.
Therapeutic Rationale
Restoring testosterone exposure within the physiologic premenopausal range represents a potential approach to addressing symptoms associated with reduced androgen activity.
We envision a future where foundational hormonal care for women fully addresses both menopause symptoms and the role of androgens in female health.
The current treatment paradigm of menopausal care needs to change.
All women will go through menopause if they live long enough.
In the US, about 55 million women are currently in some phase of menopause, with an additional 1.3 million women entering menopause each year.
All women produce testosterone, which starts to decline around the age of 30. This decline contributes to the menopausal symptoms experienced during midlife.
84% of women say their menopause symptoms interfere with their lives.
The decline of testosterone is referred to as hypoandrogenism, which is due to reduced androgen activity.
Yet, there are currently ZERO FDA-approved therapies to provide women with testosterone replacement care.
Hormonal Physiology in Women
Estrogen, progesterone, and androgens play central roles in female physiology throughout the reproductive years, influencing sexual function, mood, metabolism, musculoskeletal health, and overall well-being.
Menopause is characterized by loss of ovarian function, resulting in cessation of menses and significant changes in hormone production, including declines in estrogen, progesterone, and ovarian androgens.
Androgen Biology and Testosterone in Women
Testosterone is a physiologically relevant hormone in women, produced by both the ovaries and adrenal glands. It plays an important role in multiple biological systems, including sexual desire, energy, body composition, and aspects of mood and cognition.
Loss of ovarian androgen production, particularly following oophorectomy, results in reduced testosterone exposure, which contributes to clinically meaningful symptom burden in some women, especially in domains such as sexual function, energy, and overall well-being.

FDA Consumer Update and Approved Product Labeling (2024-2026)
Your health care provider can help you decide the best treatment based on your individual symptoms, medical history, and preferences.
Critically, the FDA's own approved drug labeling for menopausal hormone therapy products explicitly states that serum estrogen levels do not predict treatment response.
From the Minivelle (estradiol) FDA-approved prescribing information: Generally, a serum estrogen concentration does not predict an individual woman's therapeutic response to [hormone therapy]. Dosage adjustments should be based on clinical response.
The FDA initiated the removal of the boxed warnings following a comprehensive review of the scientific literature, an expert panel, and a public comment period — empowering women who have symptoms of menopause and are looking to receive potentially life-changing treatment.
